Technical Specification
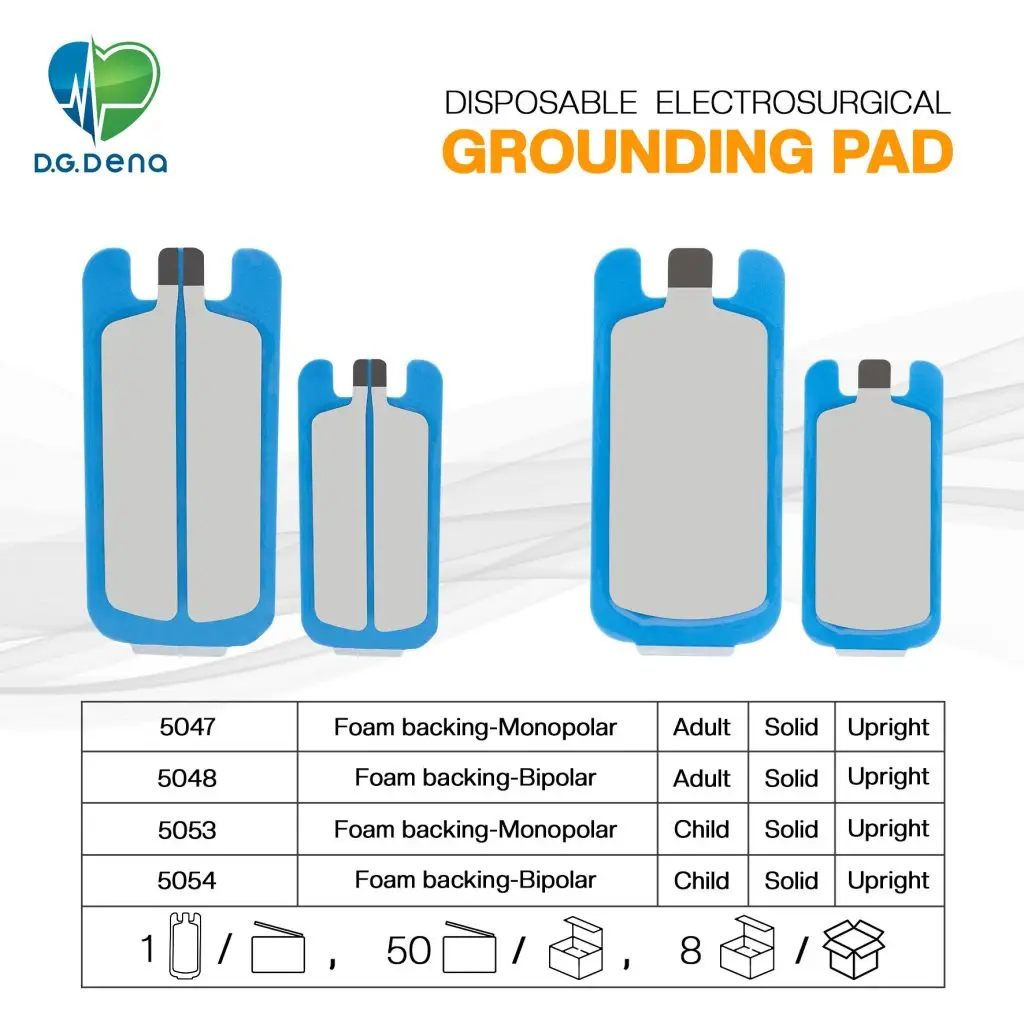
| Type |
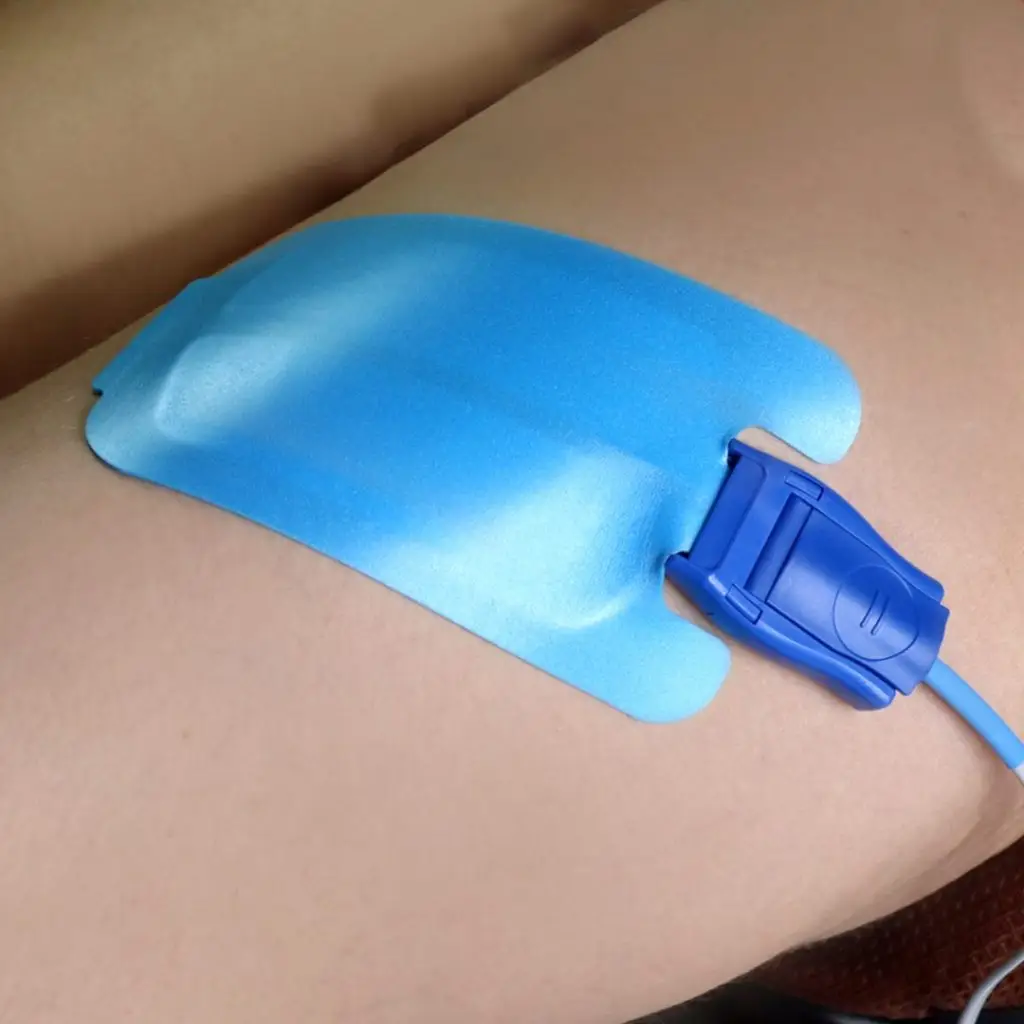
Disposable, single use, non-sterile ESU plate |
| Versions |
Non-Split & Split |
| Biocompatibility |
ISO 10993-1 for all skin contact materials |
| Performance |
Temperature rise of less than 5°C (as per ANSI/AAMI HF18:2001) |
| Backing Material |
PE Foam |
| Total Area |
Adult: 200 cm2 Infant: 135 cm2 |
| Gel Area |
Adult: 120 cm2 Infant: 76 cm2 |
| Storage Temperature |
+5°C to + 40°C |
| Shelf Life |
24 Months |
Materials
- Backing material: Waterproof flexible white PE foam, closed cell, with biocompatible acrylic adhesive
- Conductive area: Aluminum / PET film with acrylic-based hydrogel, biocompatible
- Hydrogel thickness: 0,69mm gel + aluminum support
- Release liner: PET
- Pouch: P1024ET – aluminum – PE film
- Service Box: Carton
Biocompatibility
- ISO 10993-5: No Cytotoxic
- ISO 10993-10: No Sensitizing
- ISO 10993-10: Negative intracutaneous irritation test
Conclusion: The product is biocompatible.
Packaging
- Unitary pouch in 50 units service box.
Regulatory information
D.G.Dena guarantees that this product is in conformity with Regulation (EU) 2017/745 and that it has been manufactured following the directives of the Quality Assurance System certified as ISO 13485.
This product is classified as:
- Class IIb product according to Annex VIII of Regulation (EU) 2017/745., rule 9.
- GMDN code: 58494 – Electrosurgical return electrode, single-use, non-sterile.
- EMDN code: K020102 (Electrosurgery pads (neutral electrodes) and cables, single-use)